The Hidden Cost of Compliance
Food operations have come to a critical realization: supplier issues rarely begin with a pathogen, or a labeling error, or a foreign material. They begin well before that. Supplier issues begin with a reliance on static documents.
For years, many organizations have built entire departments around a singular, comforting idea: If a supplier provides the right PDFs, the ingredient must be safe. This approach treats supplier approval like a box to be checked in a shared folder. However, the status of a certificate does not indicate the ongoing performance of a supplier. As many operations have discovered, being PDF rich often leaves an organization insight poor. Most supplier programs only escalate after an incident has already occurred.
Documents received from suppliers typically skim the surface and provide limited visibility. But, it’s rare that suppliers hide critical information. When incidents occur, the data is often not recorded in the first place. The entire purpose of supplier management is to identify and control for risks that suppliers haven’t found or managed themselves.
That’s why having a supplier’s document is not the same as having control.
The Iceberg Effect
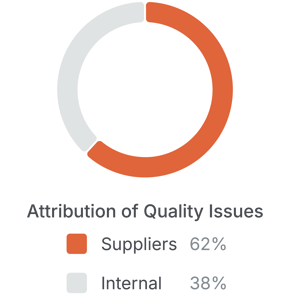
Supply chains demand attention because they account for the majority of operational deviations. In a survey of 750 quality leaders, 52% of respondents linked half of recalls to suppliers, and 62% of quality incidents were linked to supplier failures.
While these results are stark, they are not surprising. Supply chains naturally compound the number of efforts necessary to achieve control. The FDA calls this the “Iceberg Effect,” where a single internal manufacturing process may be dwarfed by 200 to 2,000 equivalent activities upstream.
It’s time to mitigate the disproportionate risk that suppliers pose. Reactive programs are costing brand integrity, consumer safety, and customer relationships.
The Maturity Gap in Supplier Management
To understand where an organization stands, it's helpful to view the evolution of supplier management through three distinct stages of maturity:
• 101: The Compliance Trap. In entry-level programs, supplier approval is a static, annual event. Documents are collected, stored, and essentially forgotten until a crisis occurs. Shallow data and subjective risk categories leave supplier risk uncontrolled.
• 201: The Silo Issue. In more mature programs, extensive supplier data exists, but it’s fragmented across disjointed folders and unintegrated software. CoAs, complaints, audit reports, and FSQA records remain entirely disconnected. Without unified data, reporting lags, so teams still fly blind.
• 301: The Comprehension Gap. In the most sophisticated operations, supplier data inputs are consolidated, but the output isn’t intuitive or actionable across teams. Quality leaders talk in plate counts, operators talk in shipments, and executives talk in costs. Without an analytical model to translate high volumes of data into a concise narrative, the organization misses the forest for the trees.
No matter which stage an organization is in, the result is the same: firefighting.
The Path to Connected Supplier Insight
The industry is currently undergoing a fundamental shift, moving from a culture of reactive compliance toward a vision of Connected Supplier Insight. Organizations successfully close the gap by following a three-step progression:
1. Onboard & Standardize: Establish a foundation by managing supplier document uploads, questionnaires, and audits within a single system. A clean list of approved suppliers is an easy result.
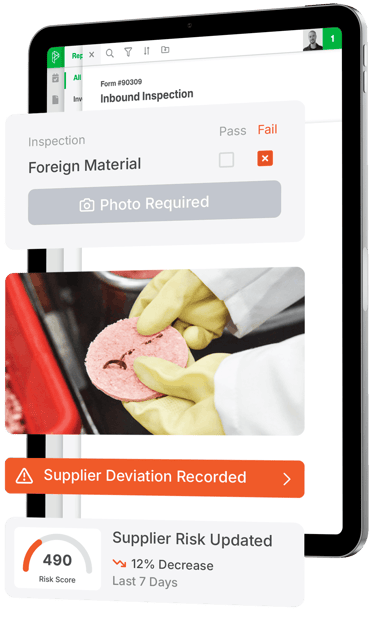
2. Connect the Floor: Integrate internal FSQA records that tag suppliers. Data from inbound inspections, product holds, swabs, and complaints will provide continuous frontline insight into actual supplier performance. This step will provide the fuel to eliminate blind spots.
3. Scale Insight: Implement Risk Scoring to make results intuitive and actionable, turning supplier data one cohesive message for all levels of the team.
Turning Data Into Action
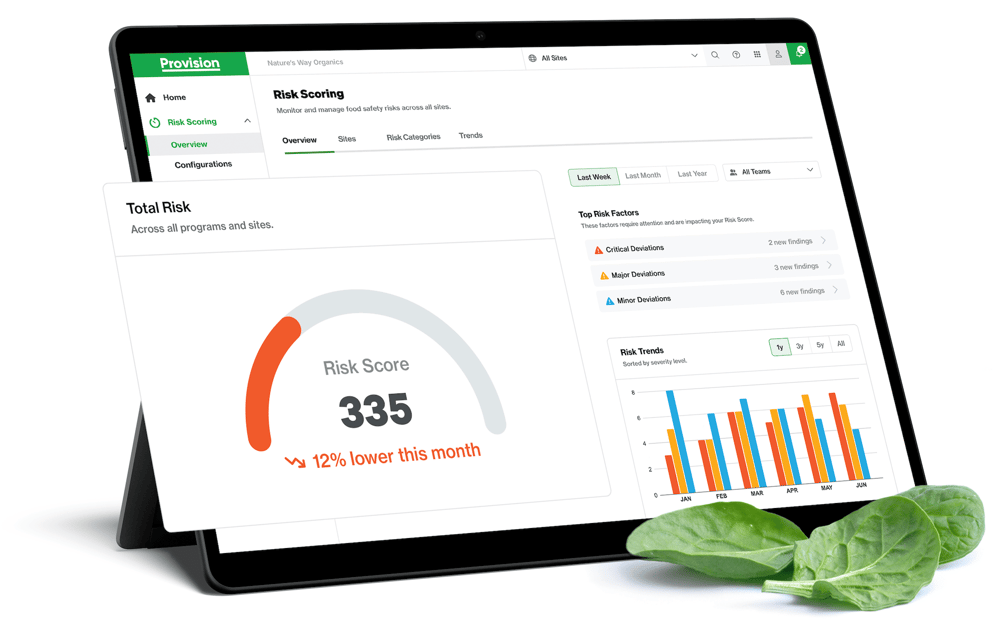
Risk Scoring works like a credit score for supply chains. By utilizing a system that continuously measures risk, teams can answer three critical questions:
1. “Is supplier risk getting better or worse?”
By objectively measuring supplier risk, teams gauge the urgency of their response and verify the effectiveness of corrective actions.
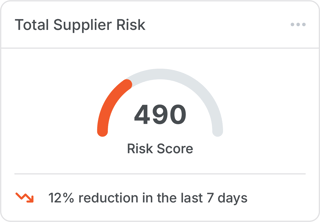
2. “Which supplier poses the greatest risk?”
Risk scoring benchmarks lists of approved suppliers, allowing procurement and quality teams to know exactly who to prioritize for audits or replacement.
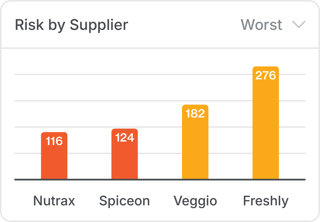
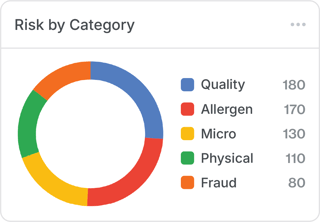
3. “Where do we point our team’s attention?”
By stack-ranking specific categories of supplier risk (for example, allergen, microbial, or physical hazards), organizations ensure that their improvement efforts are direct and intentional.
Building Operational Resilience
The shift to Connected Supplier Insight is not merely about maintaining compliance. It's a move toward operational resilience.
When visibility flows seamlessly from the plant floor to the boardroom, organizational culture changes, unifying efforts across teams. Receiving staff are empowered to better manage inbound shipments, while leadership gains the ability to make proactive, informed investments. The ultimate goal is to evolve from reactive post mortems to predictive, controlled outcomes. By treating supplier performance as a living, breathing metric, organizations can reduce risk before issues emerge. It is time to move beyond the constraints of static documents, and manage supply chains with the data-driven insight that modern brand protection requires.